 
From 27 January to 5 February 2021, an anonymous questionnaire was proposed on site to each participant just before the second dose of vaccine (between 21 and 28 days after the first one). Eligible subjects were healthcare workers vaccinated with one dose of Pfizer-BioNTech COVID-19 vaccine. We conducted a prospective and observational, single center study in university tertiary care hospital in Besançon, France. We assessed the side effects after the first dose of Pfizer-BioNTech vaccine, comparing patients with or without history of COVID-19. 
However, patients with a previous clinical or microbiologic diagnosis of Covid-19 were excluded. 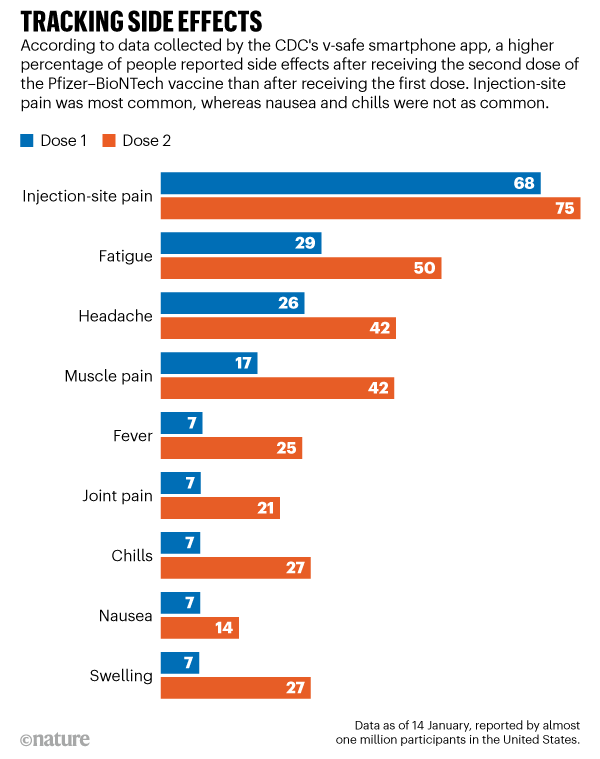
In phase 1/2/3 of the studies conducted by Pfizer-BioNTech, in vaccine recipients, reactogenicity symptoms were frequent, mostly mild to moderate, commonly reported after the second dose. Pfizer-BioNTech, an ARN vaccine, was the first recommended in France for over-50-years-old-healthcare-workers, or with high risk of severe COVID-19 and for patients over 75 years of age. The national COVID-19 vaccination program began in France at the end December 2020. Currently, only corticosteroid and tocilizumab have so far proved to be effective in patients with severe forms of COVID-19, and vaccination seems to be the most pertinent pharmacologic public health measure to control the pandemic. The coronavirus disease 2019 (COVID-19) pandemic continues to affect thousands people in France and is responsible, to date, for nearly 85 000 deaths. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
